

Physical services
Overview
During the SPEnCER project (04/2022 – 11/2023), an interdisciplinary collaboration was established between Comenius University in Bratislava (Jessenius Faculty of Medicine in Martin) and the SMEs NOVO Technologies and HOFITECH.
The project focused on spectroscopic methods for the early, non-invasive, real-time identification of selected diseases through the analysis of volatile organic compounds (VOCs) in biological samples.
Building on the technical and methodological foundations created under SPEnCER, the collaboration continued within the TEF-Health framework with the objective of developing a portable, cost-effective diagnostic device for non-invasive cancer detection based on VOC analysis in urine samples. A strong emphasis was placed on integrating artificial intelligence into signal processing and data interpretation.
The result is a functional laboratory prototype (TRL 5) that combines GC-IMS (Gas Chromatography–Ion Mobility Spectrometry) with machine learning algorithms to translate complex chemical fingerprints into probability-based diagnostic outputs. The initiative demonstrates how coordinated cooperation between academia and SMEs can advance AI-supported medical technologies toward structured clinical validation and future market deployment.
Impact
The collaboration led to clear technological and methodological advancements.
From a maturity perspective, the solution progressed from experimentally validated principles (TRL 3–4) to a functional prototype tested under laboratory conditions (TRL 5). The system is currently used for experimental measurements of urine samples from patients with various types of carcinoma in a controlled environment.
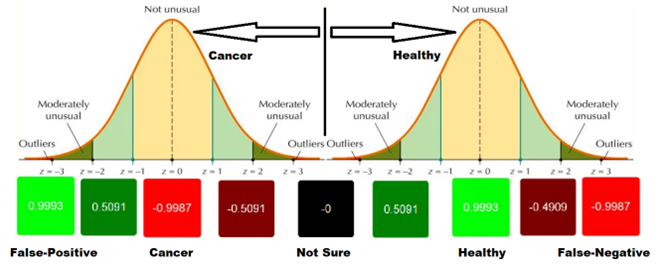
The AI component was developed to process multidimensional chromatographic outputs and classify samples as likely healthy or likely cancer. Despite limited availability of cancer samples, the model achieved:
• 88% cancer detection accuracy (29 out of 33 cases),
• 98% healthy classification accuracy (51 out of 52 cases),
• Approximately 94% overall test success across 85 samples.
Due to data scarcity, only 20% of the dataset was used for training and 80% for testing, the inverse of standard machine learning practice. To address this constraint, the team introduced an adaptive training approach (“Equity Training Strategy”), allocating greater learning effort to more complex samples.
The results, presented in Brussels in October 2025, confirmed the feasibility of combining analytical chemistry and artificial intelligence into an integrated diagnostic workflow. The project establishes a concrete basis for further dataset expansion, technical optimization, and progressive clinical verification.
The Challenge
The early detection of cancer remains a critical objective in healthcare, yet translating biochemical research into practical diagnostic tools presents significant challenges.
Cancer cells alter metabolic pathways and produce volatile and semi-volatile compounds that can be detected in bodily fluids such as urine. However, identifying and interpreting these chemical signatures requires highly sensitive instrumentation and advanced data processing methods. The project faced several technical and methodological constraints:
• Limited and imbalanced datasets, with fewer cancer samples than healthy samples.
• Variability in sample collection, storage, freezing, and transport conditions.
• The need to stabilize measurement parameters within a sensitive GC-IMS system.
• The challenge of converting complex two-dimensional chromatograms into clinically interpretable outputs.
In medical AI development, data quality, robustness, and validation under real-world variability are often more demanding than algorithm design itself. Addressing these issues was essential to move beyond proof-of-concept toward a structured prototype suitable for further validation.

The Solution
The developed device integrates a controlled VOC sensor system with AI-supported analytical software into a coherent diagnostic platform.
At hardware level, the system ensures controlled dosing and regulation of the carrier gas, stable measurement conditions, and automated decontamination between measurements. The setup includes electronic control units, sensor modules, and a regulated gas flow system designed to ensure reproducibility and reliability.
At software level, GC-IMS measurements generate two-dimensional chromatograms representing the chemical fingerprint of each sample. These multidimensional sensor responses are processed using advanced statistical methods and a feed-forward neural network that outputs probability-based classifications. Results can be visualized in a simple format (e.g., green for likely healthy, red for likely cancer), while the underlying computation remains data-driven and algorithmically robust.
Ongoing activities focus on expanding the patient database, improving robustness against sample-handling variability, identifying the compounds that most influence model decisions, and enhancing explainability. These steps aim to progressively increase the technology readiness level and prepare the system for systematic clinical verification.
Overall, the project demonstrates how research foundations established under SPEnCER, combined with continued development within TEF-Health, can translate interdisciplinary expertise into a structured, AI-supported diagnostic prototype with clear potential for further clinical and market-oriented development.
